Xanthan Gum
Xanthan gum is a nature-derived, high-molecular weight polysaccharide produced by the microorganism Xanthomonas campestris by microbial fermentation. It is highly versatile in personal care applications as it is resistant to enzymatic degradation, extremely stable over a wide range of temperatures and pHs and also in high concentrations of alcohol and salt. Xanthan gum is primarily used as a thickener, but is also the most efficient stabilizer for suspensions, emulsions, foams and solid particles in water-based formulations.
Brand name
- SATIAXANE™ xanthan gum
Cargill’s xanthan gum is available in various degrees of granulation to suit different applications. A clear version of xanthan gum is also available.
Cargill can supply grades of xanthan gum that meet the specifications of Ph.Eur. and USP/NF, when tested accordingly.
Applications
Due to the unique rheological and synergetic properties of its aqueous solutions, xanthan gum is used as a suspending agent, an emulsion and foam stabilizer, a foam enhancer, and as a thickener.
- Skin care: lotions, creams, face masks and packs
- Sun care: sun tan lotions and sunscreens
- Bath and shower care: shower gels and body washes
- Color cosmetics: make-up, mascaras and eyeliners
- Hair care: shampoos and conditioners
- Oral care: toothpaste - xanthan gum offers superior water binding properties and excellent formulation stand up in a broad range of toothpastes. It can be used as a stand alone binder or in combination with other commonly used binders
- Shaving foams
Functionalities
- Cold water soluble: The presence of anionic side chains on the xanthan gum molecules enhances hydration and makes xanthan gum soluble in cold as well as hot water.
- Viscosity control: The viscosity of xanthan gum is stable at low pH values and at high temperatures for a long period of time, whereas other hydrocolloids lose their viscosity under the same conditions.
- Temperature and acid stability: Solutions of xanthan gum are not generally affected by changes in pH or temperature and will dissolve in most acids or bases.
- Salt tolerance: The viscosity of xanthan gum is not affected by the addition of large amounts of salt and demonstrates excellent compatibility in the presence of anionic, amphoteric and non-ionic surfactants. When tested in a 250/l NaCl brine, only a slight increase in viscosity could be observed.
- Freeze/thaw stability: Due to its water binding capacity, xanthan gum solutions exhibit good freeze/thaw stability.
- Compatibility with other hydrocolloids: Xanthan gum has a synergistic increase in viscosity or gel strength when used with galactomannans, particularly locust bean gum (gel formation) and guar gum (higher viscosity).
- Shear-thinning pseudoplastic rheology: The high viscosity associated with xanthan gum solutions at low shear rates enables particle suspension and prevents oil droplets from coalescing. As the viscosity drops when shear is applied, the end consumer product can be easily scooped, poured, or squeezed from its container. Once the force is removed, the solutions regain their initial viscosity almost immediately.
- Electrolyte tolerant
- Resistant to enzymatic degradation
- Forms fluid gels with unique suspension properties
- Sparkling clarity
- Process flexibility and tolerance
- Excellent suspension for insoluble solids and oil droplets
- Effective at low concentrations
- Thickening agent
- Shear reversible behavior
- Excellent emulsion and foam stabilization
- Excellent stability
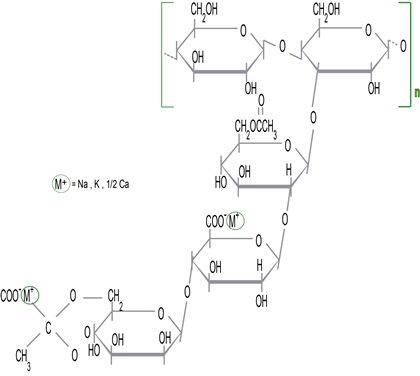
Chemistry
Xanthan gum is a hetero-polysaccharide of high molecular weight. Its main chain is constituted of glucose units. The side chain is a trisaccharide, consisting of alpha-D-mannose, which contains an acetyl group, beta-D-glucuronic acid, and a terminal beta-D-mannose unit linked with a pyruvate group.
These side chains represent about 60 percent of the molecule and give xanthan gum many of its unique properties.