Xanthan Gum
Xanthan gum is a polysaccharide widely used for its unique ability to control the rheological properties of a wide range of food products. Xanthan gum dissolves readily in hot or cold water, provides uniform brine distribution, is stable in acidic and alkaline solutions and has synergistic interactions with other hydrocolloids such as locust bean gum and guar gum.
Xanthan gum creates a pleasant texture in foods and beverages. As an ingredient in beverages, xanthan gum can produce an increase in the viscosity of a liquid. In foods like dressings and sauces, xanthan gum helps to prevent oil separation by stabilizing the emulsion. Xanthan gum as an ingredient in ice cream and other frozen foods, produces desired texture.
Xanthan gum produced by Cargill Texturizing Solutions complies with all legal, religious and safety requirements, and can be supplied and certified free of any genetically modified organism (GMO).
Applications
Bakery: Bakery & Pastry (bread type products, cake type products)
Beverages
Beverages - Alcoholic
Convenience Foods: Sauces and Dressings (emulsified sauces, tomato-based sauces, sauces with particles in suspension, dry mix sauces)
Dairy: Ice Creams and Dairy Products, Powder Products
Fruits & Vegetables: Fruit Preparations
Meat & Fish
Brands
- Satiaxane® xanthan gum
Functionality
Xanthan gum is a biopolymer.
Xanthan gum is produced as a secondary metabolite by a fermentation process, based on the culture, in aerobic conditions, of the micro-organism Xanthomonas campestris.
Xanthan gum is a hetero-polysaccharide with a very high molecular weight (between one and several million). Its main chain is composed of glucose units.
The side chain is a trisaccharide, consisting of alpha-D-mannose that contains an acetyl group, beta-D-glucuronic acid, and a terminal beta-D-mannose unit linked with a pyruvate group.
The monosaccharides present in xanthan gum are: beta-D-glucose, alpha-D-mannose and alpha-D-glucoronic acid in a ratio of 2:2:1.
The beta-D-glucoses are (1->4) linked to form the backbone. Alternate glucoses have a short, three-sugar branch consisting of a glucuronic acid sandwiched between two mannose units. Thus, the overall repeating structure is the pentasaccharide.
Structure of xanthan gum repeating unit
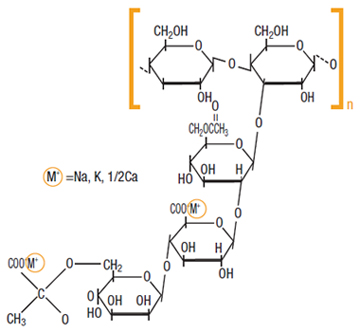
The terminal mannose can have a pyruvate group attached and the mannose adjacent to the main chain may have an acetyl group attached to C6. In general, about one branch in two has a pyruvate group, but the ratio of pyruvate to acetate varies depending on the substrain of Xanthomonas campestris used and the conditions of fermentation. The glucuronic and pyruvic acid groups give xanthan gum a highly negative charge. These acid groups are neutralized using sodium, potassium or calcium ions for food products.
In their solid state xanthan gum molecules have a helical structure. The branches fold in, to lie along the backbone.
Xanthan gum is a thickening agent. Its rigid helical structure can be melted, leading to a disorganized state with lower viscosity. The organized state is stabilized by the presence of electrolytes. The transition temperature is above 100° C in the presence of small quantities of salt.
The presence of anionic side chains on the xanthan gum molecules enhances hydration and makes xanthan gum soluble in cold water.
Xanthan gum is one of the most successful hydrocolloids due to its unique functionality, particularly in difficult environments like acid, high salt and high shear stress.
Temperature and acid stability
Solutions of xanthan gum are generally not affected by changes in pH value. Xanthan gum will dissolve in most acids or bases.
Viscosity control
The viscosity of xanthan gum is stable at low pH values and at high temperatures for a long period of time.
Salt tolerance
Viscosity is not affected by the addition of large amounts of salt; for example, in a 250 liter sodium chloride brine, only a slight increase in viscosity can be observed.
Freeze/thaw stability
Thanks to its water binding capacity, xanthan gum solutions exhibit good freeze/thaw stability.
Compatibilities
Xanthan gum is cold soluble, providing high viscosity and pseudoplastic behavior at low concentration. It can be directly dispersed in oil or a sugar solution to avoid introduction of air bubbles in water or when water is not directly available in the formulation. Dissolution takes place during processing. In order to facilitate handling, dust-free and granulated versions assist dispersion or dissolution needs.
Xanthan gum has a synergistic effect in combination with locust bean gum and konjac (gel formation) as well as with guar gum (higher viscosity). Thanks to the unique rheological and synergistic properties of its aqueous solutions, xanthan gum is used in many applications as a suspending agent and emulsion stabilizer, a foam enhancer or an improver of dough volume.
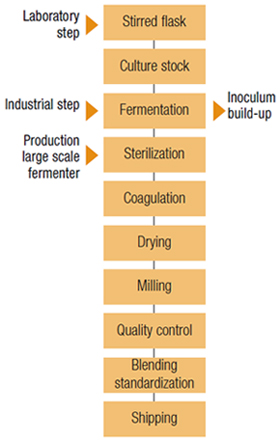
Manufacturing Process
The strain is preserved in a freeze-dried state. It is activated by inoculation into a nutrient medium containing a carbohydrate, a nitrogen source and mineral salts. After growth, the cultures are used to inoculate successive fermenters right through to the industrial scale.
Throughout the fermentation process, pH, aeration, temperature and agitation are monitored and controlled.
Once the carbohydrate is exhausted, the broth is sterilized. Then, the fermenter is emptied, cleaned and sterilized before the next fermentation takes place.
Xanthan gum is recovered by precipitation in alcohol (isopropyl or ethanol). The coagulum obtained is separated, rinsed, pressed, dried and ground before quality control.