Sweetness Explained
Many types of sugars are found in the daily diet and can exist in crystalline form or as syrups. These include for example fructose, sucrose, lactose, glucose syrup and invert sugar syrup. When the term ‘sugar’ is used, one is usually referring to ‘sucrose’, such as table sugar, white sugar, crystal sugar and brown sugar.
A few key metrics you need:
Relative Sweetness
In carefully controlled studies with good statistical treatment, objective measures of sweetness can be obtained. Relative sweetness is defined as the sweetness in watery solution in comparison to a standard sucrose solution of the same concentration. The relative sweetness for sucrose is set at 100.
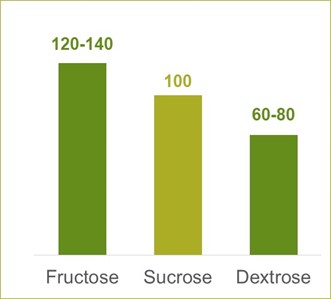 Several factors influence the relative sweetness such as concentration, temperature, viscosity etc. Therefore, figures stated in literature usually show some variance in numbers, as shown in the example in the graph.
Several factors influence the relative sweetness such as concentration, temperature, viscosity etc. Therefore, figures stated in literature usually show some variance in numbers, as shown in the example in the graph.
In applications, consideration of both flavor masking and flavor enhancement should be taken into account. As an example, fructose has a higher initial intensity of sweetness than sucrose, but it will fade away more rapidly. As a consequence, other flavors in the system will be perceived more clearly after the peak of the sweetness decreases.
Degree of Polymerization (DPx)
Carbohydrates are classified by their degree of polymerization (DPx), which is an indication of the length of the carbohydrate chain. The term sugars refers to the sum of all the mono- and di-saccharides (DP1 & DP2). Higher DPs are not classified as sugars, but as carbohydrates.
- Mono-saccharides (DP1): Simple sugars with only 1 sugar unit, such as glucose (dextrose), fructose and galactose
- Di-saccharides (DP2): Contain 2 sugar units linked to each other by a chemical bond, such as sucrose (=1 glucose and 1 fructose), maltose (2 glucose units), and lactose (1 glucose and 1 galactose)
- Tri-saccharides (DP3): Composed of 3 bound sugar units, such as maltotriose (3 glucose units)
- Oligo-saccharides (DP10-): Contain up to 9 sugar units bound to each other
- Poly-saccharides (DP10+): Contain 10 or more sugar units bound to each other, complex carbohydrates such as starch
Dextrose Equivalent (DE)
This is a measure of the degree of starch break down (hydrolysis), and is used as a general way of identifying different glucose and glucose-fructose syrups. However, different combinations of sugars can give the same DE value. Next to sweetness, below properties vary with the degree of hydrolysis:
Fermentability
Determined by the availability of fermentable sugars, with monosaccharides being readily fermentable by yeast in most applications. The applications in which fermentability is most needed are bakery and beer brewing. The advantage of glucose syrups is the possibility of modifying the degree of fermentation and the speed at which the sugars are fermented.
Freezing point depression factor (FPDF)
The effect of freezing point depression is related to the molecular weight and the effect increases toward the monosaccharide dextrose. By selecting the correct type of glucose syrup, glucose-fructose syrup or dextrose it is possible to influence the melting behavior of ice-cream. Also the keeping qualities or the direct consumption out of the freezer can be “designed” by the choice of sugar composition.
Hygroscopicity and water activity
Hygroscopicity is a measure of moisture absorption ability. Hygroscopicity is important for shelf-life as it influences enzymatic activity, the Maillard reaction, fat oxidation and microbial stability.
Inhibition of crystallization
All starch hydrolysates exert an influence on the crystallization of sweeteners in solution. Crystal formation is directly related to the saturation points of the various substances in solution. These are largely determined by molecular weight, temperature and the presence of other substances that may lower or raise total solubility and the mobility of molecules of the saccharide e.g. sucrose, in order to form the crystal. The important characteristic of glucose syrup is the ability to inhibit crystallization.
Non-enzymatic browning (Maillard reaction)
The desired browning effect in certain food applications is achieved through a condensation reaction between amino compounds and reducing sugars resulting in the formation of melanoid pigments. With the choice in sugar profile, the strength of the Maillard reaction can be controlled.
Viscosity
Viscosity is a measure of the internal friction resistance that must be overcome to make a liquid flow. Viscosity, while strongly influenced by dry substance content and temperature, is intrinsically determined by composition. The glucose viscosity is measured in “milli Pascal seconds” (mPa.s) or centipoises (cp). By a careful selection of syrups with specific viscosity, the glucose syrup can bring bulk, body & mouthfeel to food systems and also offer processing benefits and influence food products’ stability.
In this section
At Cargill, we offer the broadest range of food energy options on the market.
-
Related Information
- Sweetness Explained
- Sugar Reduction
- Partnering with Cargill
Some Cargill products are only approved for use in certain geographies, end uses, and/or at certain usage levels. It is the customer’s responsibility to determine, for a particular geography, that (i) the Cargill product, its use and usage levels, (ii) the customer’s product and its use, and (iii) any claims made about the customer’s product, all comply with applicable laws and regulations.

